Abstract
Background: Management of CML using TKIs is often constrained by treatment resistance, which portends a poor prognosis. Treatment failure may be due to therapeutic resistance (BCR-ABL1 mutation-dependent or independent), intolerance, and/or suboptimal adherence. The BCR-ABL1 T315I ("gatekeeper") genotype is insensitive to first- and second-generation TKIs, while compound mutations complicate management with all TKIs (including third-generation TKI ponatinib). HQP1351 (olverembatinib) is a novel, third-generation, orally active BCR-ABL1 TKI with evidence of antitumor activity against CML regardless of genotype (Ren X et al. Med Chem 2013;56:879-94) and a preliminary favorable safety profile in clinical trials (Jiang Q et al. Blood 2020;136:50-1).
Methods: This Chinese, open label, multicenter, phase 1 trial evaluated the safety and efficacy of olverembatinib in adults with CML in chronic phase (CML-CP) or accelerated phase (CML-AP). Eligible patients have CML-CP or CML-AP that is resistant or intolerant to first- or second-generation TKIs. Patients with severe cardiovascular diseases, hypertension, and pulmonary arterial hypertension were excluded. Olverembatinib is orally administered once every other day in 28-day cycles at 11 dose cohorts ranging from 1 to 60 mg. This study reports data on patients with long-term follow-up.
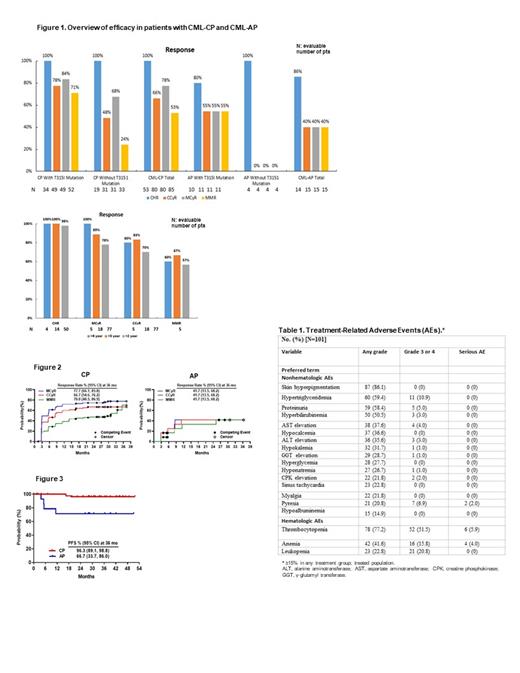
Results: From October 26, 2016, through February 2, 2021 (data cutoff date), 101 patients with CP-CML (n = 86) and AP-CML (n = 15) were enrolled and treated with olverembatinib. Seventy-one (70.3%) patients were male, the median age was 40 (range, 20-64) years, and median (range) interval from diagnosis to initial olverembatinib treatment was 6.0 (0.3-15.2) years. Eighty-four (83.2%) patients received ≥ 2 prior lines of TKI-therapy, and 63 (62.4%) harbored T315I mutation. At baseline, compound mutations were detected in 11 (10.9%) patients, of whom 7 (63.6%) had the BCR-ABL1 T315Igenotype. A total of 20 (19.8%) patients had 2 (n = 13) or ≥ 3 (n = 7) mutations. The median follow-up was 30.8 (1.2-51.8) months. As of the data cutoff date, 81 (80.2%) of 101 patients continued on treatment and 20 (14 CP-CML and 6 AP-CML) discontinued because of disease progression, intolerance, or occurrence of a secondary cancer. The cumulative median (range) drug exposure was 13,635 (1,650-20,975) mg. Of 101 patients, 18 (17.8%) were treated for > 3 years and 5 (5%) for > 4 years. Of evaluable patients without baseline responses, 97.0% had complete hematologic responses (CHR), 62.1% complete cytogenetic responses (CCyR), and 51.0% major molecular responses (MMR). Most evaluable patients with T315I mutations experienced 100% for CHR, 83.7% for MCyR, and 71.2% for MMR among patients in CP-CML, as well as 80.0% for CHR and 54.5% each for MCyR and MMR in AP-CML. At 36 months, the PFS rate (95% CI) was 96.3% (89.1%-98.8%) in patients with CP-CML and 71.4% (40.6%‒88.2%) in those with AP-CML. Treatment responses were durable and unaffected by baseline BCR-ABL1 mutational status. Corresponding values in patients with > 4 years of treatment were 100% (CHR), 80% (CCyR), and 60% (MMR). The mean (95% confidence interval) PFS rate was 100% (100%-100%) at 36 months, 100% at 48 months, and not reached (NR-NR) at 60 months. Most treatment-related adverse events were grade 1 or 2. The most frequent nonhematologic adverse event was (mostly grade 1 or 2) skin hyperpigmentation (86.1%). Grade ≥ 3 nonhematologic AEs included hypertriglyceridemia (10.9%), pyrexia (6.9%), and proteinuria (5.0%). The most common hematologic treatment-related adverse event was thrombocytopenia in 78 (77.2%) patients, including 52 (51.5% of total population) with grade ≥ 3 and 6 (5.9%) with serious adverse events. Leukopenia was grade ≥ 3 in 21 (20.8%) patients but not serious, while anemia was grade 3 or higher in 16 (15.8%) patients and serious in 4 (4.0%).
Conclusions: In patients with TKI-resistant CML-CP or CML-AP and long-term treatment, olverembatinib was efficacious and well tolerated. Internal study identifier HQP1351-SJ002.
Chen: Ascentage Pharma (Suzhou) Co., Ltd: Current Employment, Current equity holder in publicly-traded company. Niu: Ascentage Pharma (Suzhou) Co., Ltd: Current Employment, Current equity holder in publicly-traded company. Men: Ascentage Pharma (Suzhou) Co., Ltd.: Current Employment, Current equity holder in publicly-traded company. Wang: Ascentage Pharma (Suzhou) Co., Ltd: Current Employment, Current equity holder in publicly-traded company. Yang: Ascentage Pharma (Suzhou) Co., Ltd: Current Employment, Current equity holder in publicly-traded company, Other: Leadership and other ownership interests, Patents & Royalties, Research Funding. Zhai: Ascentage Pharma Group Inc.: Current Employment, Current equity holder in publicly-traded company, Other: Leadership and other ownership interests, Patents & Royalties, Research Funding; Ascentage Pharma (Suzhou) Co., Ltd.: Current Employment, Current equity holder in publicly-traded company, Other: Leadership and other ownership interests, Patents & Royalties, Research Funding.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal